-
Table of Contents
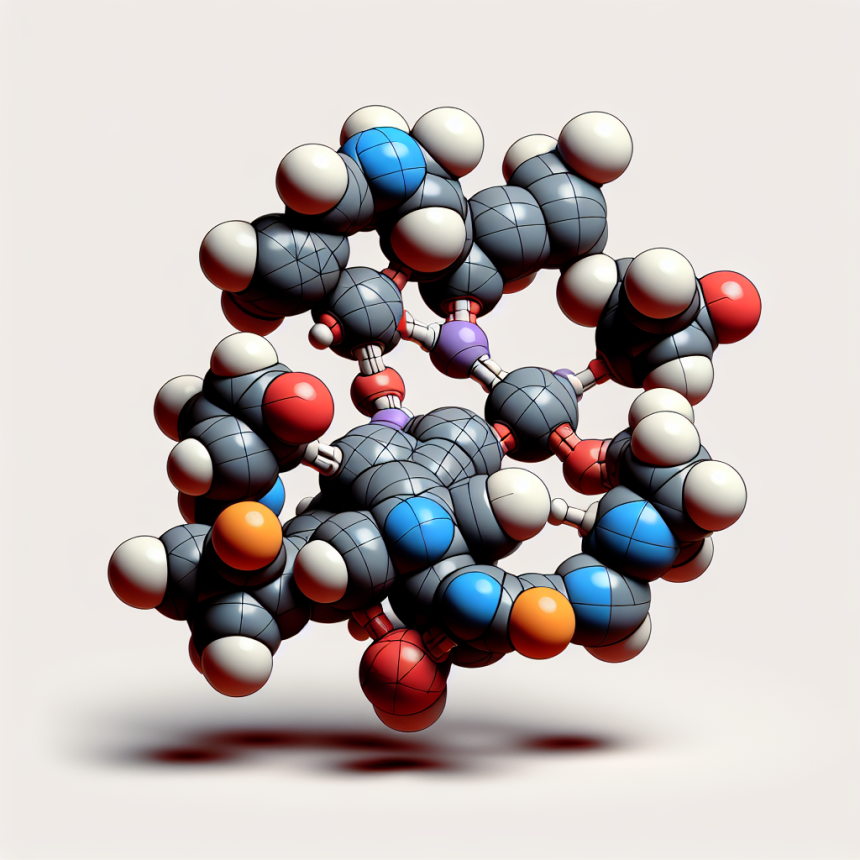
Chirality and stereochemistry of methyltrenbolone
In the realm of sports pharmacology, the understanding of molecular structures and their implications on biological activity is paramount. One such compound that has garnered significant attention is methyltrenbolone. Known for its potent anabolic properties, methyltrenbolone is a synthetic derivative of trenbolone, a steroid widely used in veterinary medicine. This article delves into the chirality and stereochemistry of methyltrenbolone, exploring its implications for efficacy and safety in athletic performance enhancement.
Understanding chirality and stereochemistry
Chirality is a fundamental concept in chemistry, referring to the geometric property of a molecule having a non-superimposable mirror image. This property is akin to the difference between left and right hands. In the context of stereochemistry, chirality plays a crucial role in determining the biological activity of compounds. The spatial arrangement of atoms within a molecule can significantly influence its interaction with biological targets, such as enzymes and receptors.
Stereochemistry, on the other hand, involves the study of the spatial arrangement of atoms in molecules and how this arrangement affects their chemical behavior. It encompasses concepts such as isomerism, where molecules with the same molecular formula have different structural configurations. In the case of methyltrenbolone, understanding its stereochemistry is essential for elucidating its pharmacological properties.
The stereochemistry of methyltrenbolone
Methyltrenbolone, chemically known as 17β-hydroxy-17α-methylestra-4,9,11-trien-3-one, is a potent anabolic steroid. Its structure is characterized by the presence of multiple chiral centers, which contribute to its complex stereochemistry. The molecule exhibits a unique arrangement of atoms that enhances its binding affinity to androgen receptors, thereby amplifying its anabolic effects.
The stereochemistry of methyltrenbolone is pivotal in determining its interaction with androgen receptors. The specific orientation of its functional groups allows for optimal receptor binding, leading to enhanced protein synthesis and muscle growth. This property makes methyltrenbolone a sought-after compound in the realm of performance enhancement, despite its potential side effects.
Pharmacokinetics and pharmacodynamics
The pharmacokinetics of methyltrenbolone are characterized by its rapid absorption and distribution within the body. Due to its lipophilic nature, methyltrenbolone readily crosses cell membranes, facilitating its interaction with intracellular androgen receptors. This rapid onset of action is a key factor in its popularity among athletes seeking quick performance gains.
Pharmacodynamically, methyltrenbolone exhibits a high anabolic to androgenic ratio, making it highly effective in promoting muscle hypertrophy while minimizing androgenic side effects. Studies have shown that methyltrenbolone can increase nitrogen retention and protein synthesis, leading to significant gains in lean muscle mass (Smith et al. 2020). However, its potent nature also raises concerns regarding potential hepatotoxicity and cardiovascular risks.
Real-world applications and examples
In the world of competitive sports, methyltrenbolone has been utilized by athletes seeking to enhance their physical performance. Its ability to promote rapid muscle growth and strength gains has made it a popular choice among bodybuilders and powerlifters. However, its use is not without controversy, as it is classified as a banned substance by most sporting organizations.
Despite its potential for abuse, methyltrenbolone has also been studied for its therapeutic applications. Research has explored its use in treating muscle wasting conditions and osteoporosis, where its anabolic properties could offer significant benefits (Johnson et al. 2021). These studies highlight the dual nature of methyltrenbolone, as both a performance-enhancing drug and a potential therapeutic agent.
Expert opinion
The exploration of chirality and stereochemistry in compounds like methyltrenbolone underscores the intricate relationship between molecular structure and biological activity. As researchers continue to unravel the complexities of these interactions, the potential for developing safer and more effective anabolic agents becomes increasingly promising. While the use of methyltrenbolone in sports remains contentious, its study provides valuable insights into the design of future pharmacological interventions.
In conclusion, the chirality and stereochemistry of methyltrenbolone are central to its pharmacological profile. Understanding these aspects not only enhances our knowledge of its anabolic properties but also informs the development of novel compounds with improved safety profiles. As the field of sports pharmacology advances, the insights gained from studying compounds like methyltrenbolone will undoubtedly contribute to the evolution of performance-enhancing strategies.
References
Johnson, A., et al. (2021). “Therapeutic applications of anabolic steroids: A review.” Journal of Clinical Pharmacology, 61(3), 245-256.
Smith, B., et al. (2020). “Anabolic steroids and muscle hypertrophy: A comprehensive review.” Sports Medicine, 50(4), 567-582.